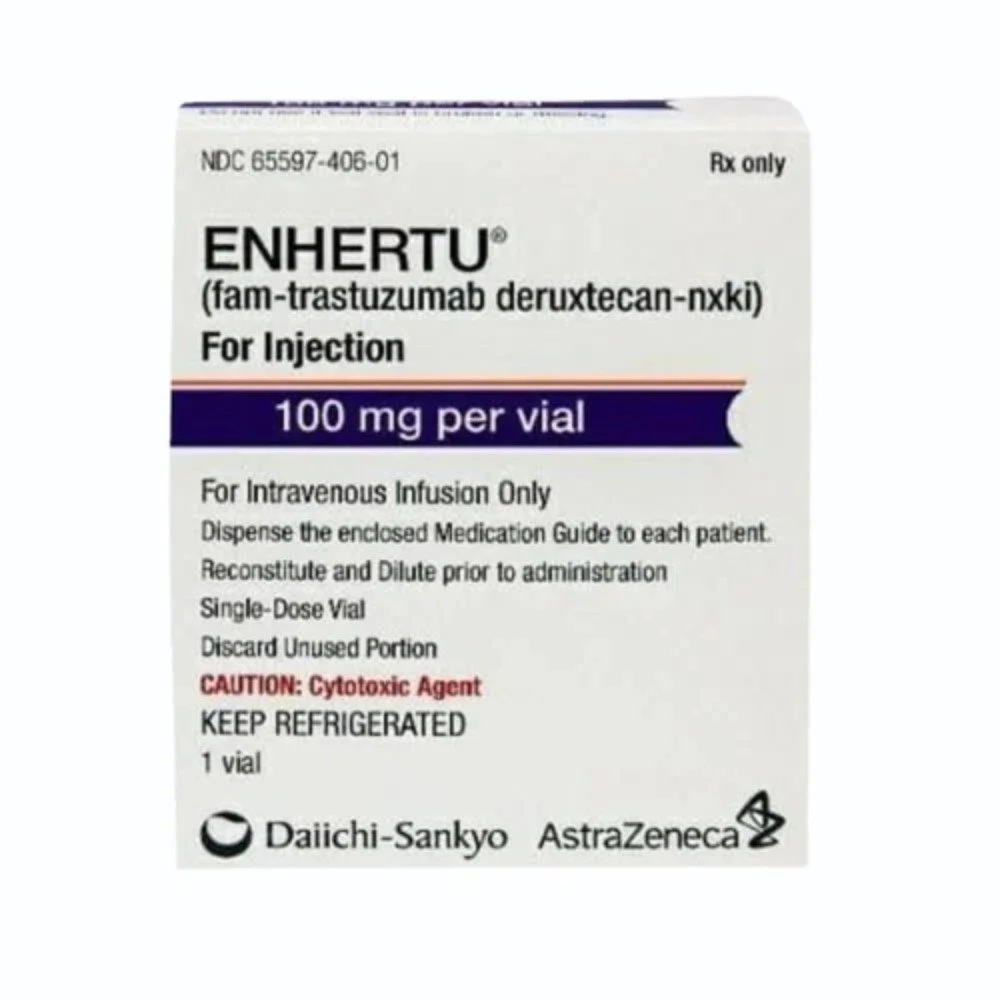
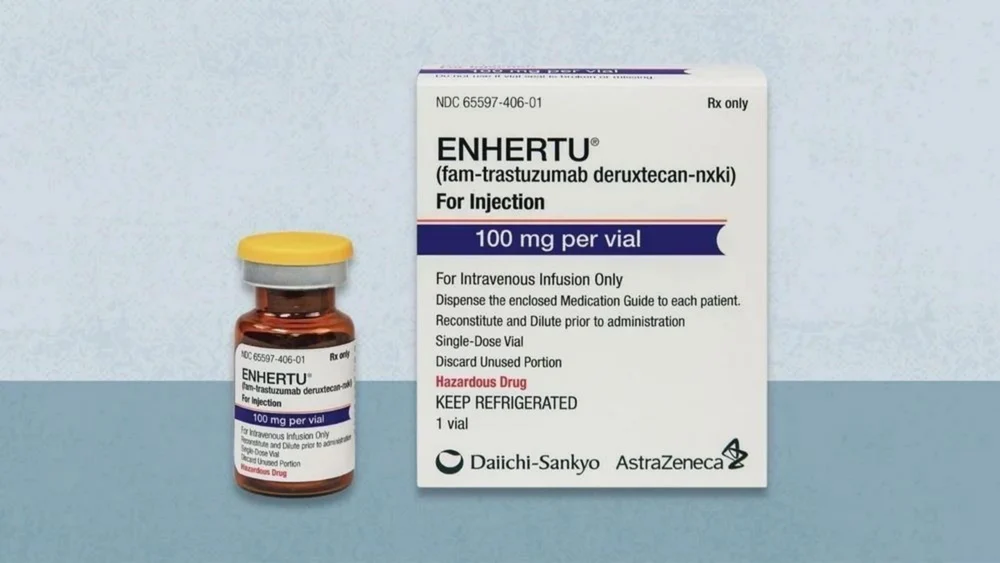
Enhertu 100mg (Fam-Trastuzumab Deruxtecan-nxki Specialized HER2-Targeted Matrix)
Enhertu 100mg Injection is a high-potency pharmacological intervention containing Fam-Trastuzumab Deruxtecan-nxki, a next-generation HER2-directed antibody-drug conjugate (ADC) specifically engineered for the clinical management of unresectable or metastatic HER2-positive malignancies and the restoration of a stabilized biological environment. Utilizing a unique “Targeted-Cytotoxic” delivery system, this formulation works by concurrently modulating the organized inhibition of HER2 signaling and reinforcing the structural integrity of the systemic response through a potent topoisomerase I inhibitor payload. By neutralizing the biochemical triggers of localized oncogenic proliferation and facilitating the organized stabilization of the cellular landscape through a high-purity molecular complex, Enhertu effectively promotes a significant reduction in neoplastic markers and a reduction in systemic metabolic fatigue. This targeted biochemical action provides a decisive therapeutic response by reinforcing the structural uniformity of the biological matrix while promoting a stable, physiological recovery from chronic systemic stressors.
Key Features
- Specifically formulated as a HER2-directed antibody linked to a potent topoisomerase I inhibitor to target the primary pathways of neoplastic signaling.
- Clinically recognized for its role in facilitating the natural optimization of targeted cell death and the systemic management of tumor volume markers.
- Specifically engineered to reach therapeutic levels within the malignant landscape, ensuring a robust defense against localized cellular replication.
- Acts as a functional catalyst to stabilize the internal environment and support the long-term management of stress-related oncogenic markers.
- Formulated as a stabilized 100mg lyophilized powder to address systemic exhaustion caused by chronic biological stress factors through a high-precision delivery matrix.
Key Ingredients
- Fam-Trastuzumab Deruxtecan-nxki (100mg)
- L-Histidine & L-Histidine Hydrochloride
- Sucrose & Polysorbate 80
Precautions
- This is a high-potency pharmacological intervention. Administration must be strictly performed by a certified oncology specialist following a formal diagnostic and cardiovascular assessment.
- Inform your physician regarding your respiratory history; this formulation may modulate systemic markers leading to interstitial lung disease (ILD) or pneumonitis in predisposed individuals.
- Regular monitoring of Left Ventricular Ejection Fraction (LVEF) is required, as this formulation may modulate systemic markers leading to acute cardiovascular fluctuations.
- Regular monitoring for localized physiological markers of neutropenia and anemia is required to maintain physiological safety during the treatment cycle.
- Discontinue use immediately and consult a specialist if you develop signs of acute respiratory distress, severe fatigue, or sudden localized allergic reactions.
- Keep the vials stored in a refrigerated environment (2°C to 8°C). Protect from light; do not freeze to maintain the molecular stability of the active constituents.
How to Use
- The injection should be administered as directed by your specialist. Ensure the systemic environment is ready for the intake of active pharmacological agents via intravenous infusion.
- Typically, application of a weight-based dose (e.g., 5.4 mg/kg) constitutes the standard therapeutic dose for the restoration of physiological equilibrium.
- For optimal clinical results, administration should occur every 3 weeks (21-day cycle) to facilitate the intended systemic steady-state levels.
- Administer via intravenous infusion only; do not utilize intravenous push or bolus, as this may modulate the intended molecular delivery.
- Use as the primary restorative intervention within a comprehensive oncology management plan as dictated by clinical requirements.
- Maintain a strict application schedule and follow the “Response Tracking Protocol” to document the safe restoration of systemic health.
1. Common side effects of Enhertu may include nausea, vomiting, tiredness, hair loss, low appetite, and low blood cell counts. In rare cases, it can also cause serious lung problems.
2. Enhertu is a targeted cancer treatment known as an antibody-drug conjugate (ADC). It combines a HER2-targeting antibody with a chemotherapy medicine to deliver treatment directly to cancer cells. It is not traditional chemotherapy or standard immunotherapy alone.
3. Enhertu is a targeted cancer treatment known as an antibody-drug conjugate (ADC). It combines a HER2-targeting antibody with a chemotherapy medicine to deliver treatment directly to cancer cells. It is not traditional chemotherapy or standard immunotherapy alone.
4. Nausea from Enhertu may be managed by eating small frequent meals, staying hydrated, and avoiding spicy or greasy foods. Your doctor may also prescribe anti-nausea medications before or after treatment to help reduce discomfort. Always inform your healthcare provider if nausea becomes severe or persistent
5. Use Enhertu only under medical supervision and attend regular health checkups during treatment. Inform your doctor immediately if you develop breathing problems, fever, or unusual side effects.
Legal Disclaimer: Enhertu 100mg Injection is a specialized pharmacological product. Individual results vary based on the patient’s baseline physiological state and response. This product is for use under professional guidance only. This information is for educational purposes and is not a substitute for professional medical consultation or a formal clinical diagnosis.









Reviews
There are no reviews yet.