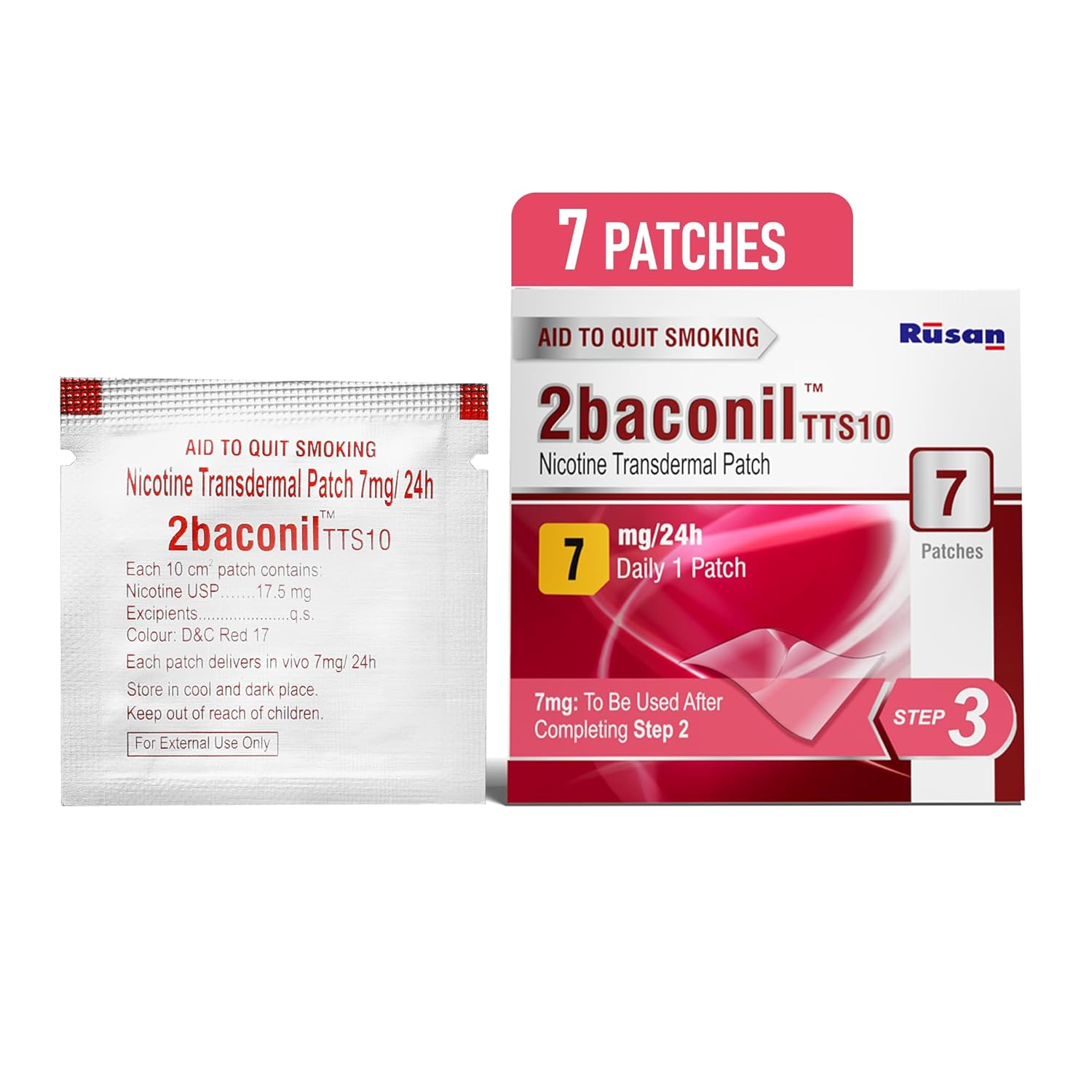
2baconil 7mg Nicotine Gums (Nicotine Polacrilex)
2baconil 7mg Nicotine Gum is a high-potency smoking cessation intervention containing Nicotine Polacrilex, a specialized nicotine-resin complex engineered for the clinical management of severe tobacco dependency. Utilizing a unique “Transmucosal-Absorption” delivery system, this formulation works by concurrently modulating the organized release of therapeutic nicotine and reinforcing the structural integrity of the neurological pathways. By neutralizing the biochemical triggers of intensive nicotine withdrawal and facilitating the organized stabilization of plasma nicotine levels, 2baconil effectively promotes a significant reduction in acute cravings and a reduction in metabolic dullness associated with cessation. This targeted biochemical action provides a decisive therapeutic response by reinforcing the structural uniformity of the cholinergic system while promoting a stable, physiological recovery from heavy tobacco-related stressors.
Key Features
- Specifically formulated with a high-density nicotine-resin complex to target primary nicotinic receptors in heavy users.
- Clinically recognized for its role in facilitating the natural optimization of cessation success by neutralizing peak withdrawal symptoms.
- Specifically engineered to reach therapeutic levels through the buccal mucosa, ensuring a robust defense against localized cravings.
- Acts as a functional catalyst to stabilize mood and support the reduction of concentration-related cessation fatigue.
- Formulated as a 7mg dose to address advanced nicotine dependency in individuals transitioning from high-frequency tobacco use.
Key Ingredients
- Nicotine Polacrilex (USP 7mg)
- Gum Base
- Sodium Carbonate
- Sorbitol & Maltitol
Precautions
- This is a high-potency cessation health intervention. Consult with a specialist prior to use if you have a history of cardiovascular disease, arrhythmias, or recent myocardial infarction.
- Do not use if you have severe dental conditions, jaw disorders (TMJ), or localized inflammation of the oral mucosa.
- Inform your physician if you have a history of severe hypertension or peripheral vascular disease before administration.
- Discontinue use immediately if persistent mouth sores, severe hiccups, or localized gastrointestinal discomfort occurs during the treatment period.
- Consult a healthcare provider before administration during pregnancy or lactation to ensure the physiological safety of the restoration cycle.
- Keep the pack tightly sealed and store in a cool, dry place (below 25°C). Protect from direct light to maintain the molecular stability of the active constituents.
How to Use
- The gum should be used whenever a tobacco craving occurs. Ensure the oral cavity is free from food or beverages for 15 minutes prior to administration.
- The frequency of administration should be strictly dictated by your clinical cessation protocol. Do not exceed the maximum daily limit as prescribed by your specialist.
- For optimal clinical results, administration should follow the “Chew and Park” technique to allow for steady-state absorption.
- Chew the gum slowly until a tingling sensation or peppery taste is established, then “park” it between the cheek and gum until the taste fades. Repeat the process for approximately 30 minutes.
- Use as part of a comprehensive cessation regimen, gradually reducing the potency or frequency of administration as dictated by clinical requirements.
- Maintain a regular application schedule to document visible improvements in behavioral markers and the restoration of systemic equilibrium.
Legal Disclaimer: 2baconil 7mg Nicotine Gums are a specialized pharmacological product. Individual results vary based on the patient’s baseline nicotine dependency and physiological response. This product is for use under professional guidance only. This information is for educational purposes and is not a substitute for professional medical consultation or a formal clinical diagnosis.












Reviews
There are no reviews yet.