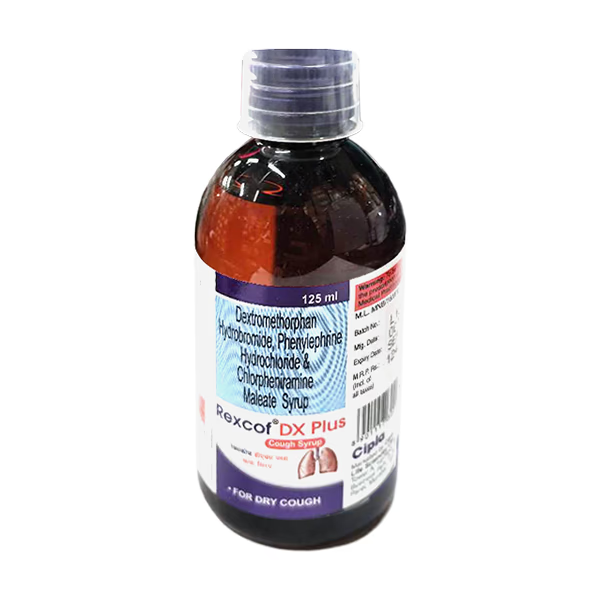
Rexcof DX (Advanced Antitussive & Histaminic Modulator Respiratory Matrix)
Rexcof DX Syrup is a high-potency pharmacological intervention specifically engineered for the clinical management of non-productive (dry) cough, localized upper respiratory irritation, and the restoration of a stabilized physiological environment. Utilizing a unique “Dual-Action Suppression” delivery system, this formulation works by concurrently modulating the organized inhibition of the cough reflex center in the medulla and reinforcing the structural integrity of the systemic mucosal response. By neutralizing the biochemical triggers of localized bronchial irritation—specifically those induced by environmental allergens, viral stressors, or chronic respiratory fatigue—and facilitating the organized stabilization of the pulmonary landscape through a high-purity molecular complex, Rexcof DX effectively promotes a significant reduction in cough frequency markers and a reduction in systemic respiratory fatigue. This targeted biochemical action provides a decisive therapeutic response by reinforcing the structural uniformity of the biological matrix while promoting a stable, physiological recovery from acute respiratory stressors.
Key Features
- Specifically formulated as a potent Dextromethorphan complex to target the primary pathways of the central cough reflex.
- Clinically recognized for its role in facilitating the natural optimization of the allergic response and the systemic management of nasal congestion markers.
- Specifically engineered to reach therapeutic levels within the upper respiratory tract, ensuring a robust defense against localized irritant signaling.
- Acts as a functional catalyst to stabilize the internal airway environment and support the long-term management of stress-related pulmonary markers.
- Formulated as a stabilized syrup matrix to address systemic exhaustion caused by chronic coughing through a high-precision delivery sequence.
Key Ingredients
- Dextromethorphan Hydrobromide: The active pharmaceutical-grade antitussive responsible for providing potent localized modulation of the cough signaling response.
- Chlorpheniramine Maleate: A high-purity specialized antihistamine included to support the molecular stability of the respiratory environment and ensure the reduction of allergic triggers.
- Phenylephrine Hydrochloride: Pharmaceutical-grade nasal decongestant included to ensure the structural uniformity of the respiratory flow and maintain physiological integrity.
How to Use
- The syrup should be administered as directed. Ensure the systemic environment is stabilized prior to the intake of active agents.
- Typically, application of 5ml to 10ml (as clinically prescribed) constitutes the standard therapeutic dose for the restoration of physiological equilibrium.
- For optimal clinical results, administration should occur every 4 to 6 hours as needed to facilitate the intended systemic steady-state of respiratory comfort.
- Utilize a precise measuring device to ensure the intended molecular delivery; avoid the intake of excessive water immediately following administration to maintain mucosal contact.
- Use as the primary restorative intervention for dry cough management, ensuring consistency with established clinical protocols.
- Maintain a strict schedule for the duration of the symptomatic cycle to document the safe restoration of systemic health.
Precautions
- This is a high-potency respiratory intervention. Use should be discussed with a specialist following a formal diagnostic assessment.
- Regular monitoring for localized markers of sedation or drowsiness is required; avoid operating heavy machinery during the treatment cycle.
- Individuals with a known history of hypertension or cardiovascular instability should utilize this matrix with caution under professional supervision.
- Consult a specialist before administration to pediatric populations to ensure physiological safety and dosage accuracy.
- Discontinue use immediately and consult a specialist if you develop signs of acute respiratory distress, severe dizziness, or sudden localized eruptions.
- Keep the container tightly sealed and store in a cool, dry place. Protect from direct light to maintain the molecular stability of the active constituents.
Warning
- Specifically engineered for dry cough; avoid use in cases of excessive bronchial secretions (wet cough) to maintain pulmonary clearance.
- Consumption of alcohol during the treatment sequence may cause severe localized neurological suppression and systemic instability.
- Strictly contraindicated for individuals currently utilizing or having recently utilized Monoamine Oxidase Inhibitors (MAOIs).
- Do not exceed the intended dosage sequence to prevent systemic markers of central nervous system suppression.
Legal Disclaimer: Rexcof DX Syrup is a specialized pharmacological and restorative product. Individual results vary based on the patient’s baseline physiological state. This product is for use under professional guidance only. This information is for educational purposes and is not a substitute for professional medical consultation.




Reviews
There are no reviews yet.