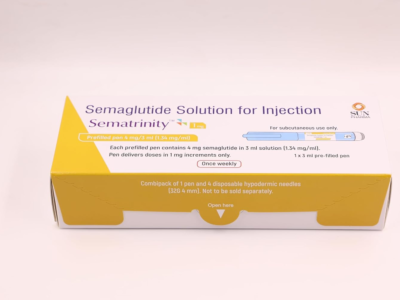
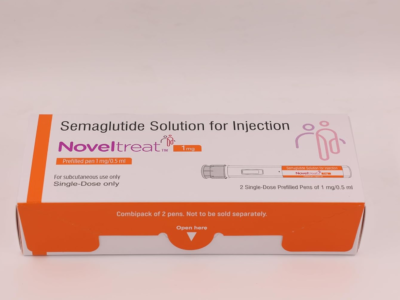
Noveltreat 0.5 mg (Specialized GLP-1 Receptor Agonist Metabolic Matrix)
Noveltreat 0.5 mg Pre-filled Pen is a high-potency pharmacological intervention containing Semaglutide, a long-acting glucagon-like peptide-1 (GLP-1) receptor agonist specifically engineered for the clinical management of Type 2 Diabetes Mellitus and the restoration of a stabilized metabolic environment. Utilizing a unique “Incretin-Mimetic” delivery system, this formulation works by concurrently modulating the organized stimulation of insulin secretion and reinforcing the structural integrity of the systemic glycemic response. By neutralizing the biochemical triggers of localized postprandial glucose excursions—and facilitating the organized stabilization of the metabolic landscape through a high-purity molecular complex—Noveltreat effectively promotes a significant reduction in HbA1c markers and a reduction in systemic metabolic fatigue. This targeted biochemical action provides a decisive therapeutic response by reinforcing the structural uniformity of the biological matrix while promoting a stable, physiological recovery from chronic metabolic stressors.
Key Features
- Specifically formulated as a potent GLP-1 receptor agonist to target the primary pathways of endogenous insulin production and glucagon inhibition.
- Clinically recognized for its role in facilitating the natural optimization of glucose metabolism and the systemic management of chronic HbA1c levels.
- Specifically engineered to reach therapeutic levels within the hypothalamic landscape, ensuring a robust defense against localized appetite dysregulation.
- Acts as a functional catalyst to stabilize the internal environment and support the long-term management of stress-related cardiovascular markers.
- Formulated as a stabilized 0.5 mg pre-filled pen to address systemic exhaustion through a high-precision, once-weekly delivery matrix.
Key Ingredients
- Semaglutide (0.5 mg)
- Disodium Phosphate Dihydrate
- Propylene Glycol & Phenol
How to Use
- The injection should be administered as directed. Ensure the targeted subcutaneous landscape is gently cleansed and stabilized prior to the intake of active agents.
- Typically, application of a once-weekly titrated dose (as per the clinical protocol) constitutes the standard therapeutic dose for the restoration of physiological equilibrium.
- For optimal clinical results, administration should occur on the same day each week, regardless of nutritional intake, to facilitate the intended systemic steady-state levels.
- Administer via subcutaneous injection in the abdominal matrix, thigh, or upper arm; rotate the application site during each sequence to prevent localized tissue strain.
- Use as the primary restorative intervention within a comprehensive metabolic management plan, including nutritional and physical activity modifications.
- Maintain a strict application schedule and follow the “Glycemic Response Tracking Protocol” to document the safe restoration of systemic health.
Precautions
- This is a high-potency metabolic intervention. Administration must follow a formal diagnostic and endocrine assessment by a certified specialist.
- Regular monitoring for localized markers of pancreatic inflammation is required; discontinue use if acute abdominal distress occurs.
- When utilized in conjunction with other high-affinity insulin secretagogues, monitor for systemic glucose oscillations to maintain physiological safety.
- Regular assessment of renal markers is advised, particularly during the initiation cycle where gastrointestinal oscillations may occur.
- Discontinue use immediately and consult a specialist if you develop signs of acute thyroid nodules, persistent nausea, or sudden localized eruptions.
- Keep the pre-filled pen stored in a refrigerated environment (2°C to 8°C) before first use. After initiation, the pen may be maintained at room temperature (below 30°C) for up to 56 days.
Warning
- Individuals with a personal or family history of Medullary Thyroid Carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) are strictly contraindicated.
- Severe gastrointestinal events, including nausea and vomiting, may occur during the titration phase; maintain hydration to prevent secondary renal strain.
- Rapid improvement in glucose control may lead to temporary oscillations in diabetic retinopathy markers; regular ophthalmic screening is mandatory.
- Strictly for subcutaneous administration; avoid intravenous or intramuscular delivery to maintain the intended molecular delivery sequence.
Legal Disclaimer: Noveltreat 0.5 mg Pre-filled Pen is a specialized pharmacological product. Individual results vary based on the patient’s baseline physiological state. This product is for use under professional guidance only. This information is for educational purposes and is not a substitute for professional medical consultation.








Reviews
There are no reviews yet.