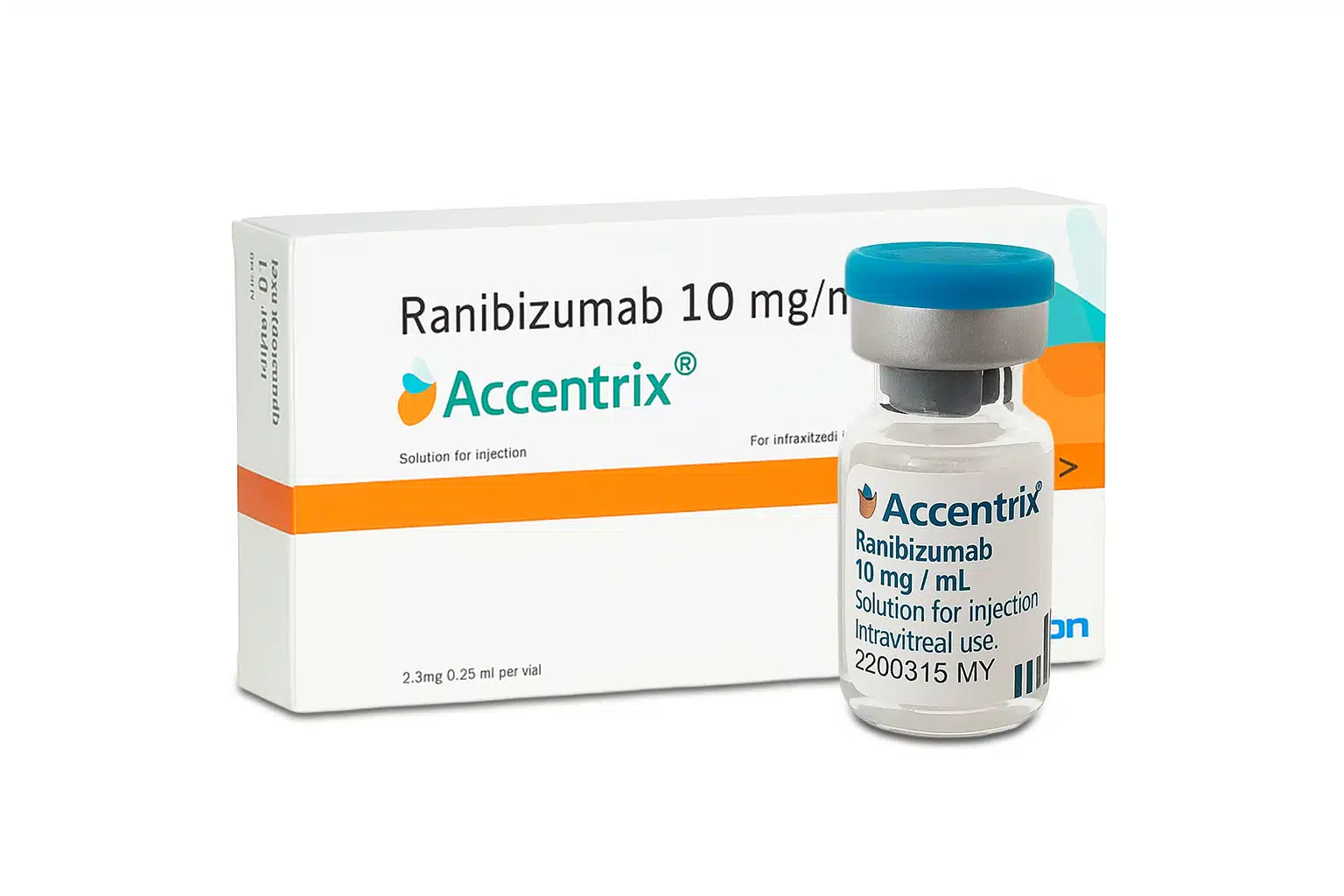
Accentrix Solution for Injection
Accentrix is a high-potency ophthalmic intervention containing Ranibizumab (10 mg/ml), a specialized recombinant humanized monoclonal antibody fragment. Specifically engineered for the clinical management of neovascular (wet) age-related macular degeneration (AMD) and other retinal vascular disorders, Accentrix works by targeting the biological drivers of abnormal blood vessel growth. Ranibizumab acts as a potent vascular endothelial growth factor (VEGF) inhibitor, specifically binding to the receptor-binding site of active forms of VEGF-A. This targeted biochemical action provides a decisive therapeutic response to suppress the leakage of fluid and blood into the retina, effectively reducing macular edema and stabilizing or improving visual acuity in patients with sight-threatening conditions.
Key Features
- Specifically binds to and neutralizes VEGF-A to prevent the growth of leaky, abnormal blood vessels.
- Effectively resolves the swelling in the central part of the retina to restore visual clarity.
- Clinically proven to stabilize vision and, in many cases, provide significant gains in letter-reading ability.
- Formulated for direct injection into the vitreous cavity for localized therapeutic action.
- The small antibody fragment size allows for efficient penetration through the retinal layers.
Key Ingredients
- Ranibizumab (10 mg/ml)
- α,α-Trehalose Dihydrate
- L-Histidine
- Polysorbate 20
- Water for Injection
Precautions
- Must be administered only by a qualified ophthalmologist experienced in intravitreal injections.
- Intravitreal injections are associated with a risk of endophthalmitis (serious eye infection); report any pain, redness, or light sensitivity immediately.
- Transient increases in eye pressure are common after injection; your specialist will monitor this during the procedure.
- There is a small risk of retinal tears or detachment following the procedure; any sudden “floaters” or flashes of light require urgent evaluation.
- Consult your healthcare professional before use, as systemic exposure, though low, should be minimized.
- Do not drive or operate machinery immediately after injection, as your vision may be temporarily blurred or pupils may be dilated.
How to Use
- Accentrix is administered as a single intravitreal injection into the affected eye under sterile conditions.
- Local anesthetic drops or a small injection will be used to ensure the procedure is comfortable and pain-free.
- Typically administered once a month initially, with the interval adjusted based on visual acuity and anatomical outcomes (Pro Re Nata or Treat-and-Extend protocols).
- Use any prescribed antibiotic or anti-inflammatory eye drops exactly as directed by your surgeon.
- Regular follow-up appointments involving Optical Coherence Tomography (OCT) scans are mandatory to track the treatment’s progress.
- Avoid rubbing your eye or exposing it to contaminated water (swimming pools/hot tubs) for several days following the injection.
Legal Disclaimer: Accentrix Solution for Injection is a prescription-only pharmaceutical product. Individual results may vary based on the duration and severity of the retinal condition. This product is not intended for the treatment of non-vascular eye diseases. This information is for educational purposes and is not a substitute for professional medical consultation from a retina specialist.




Reviews
There are no reviews yet.