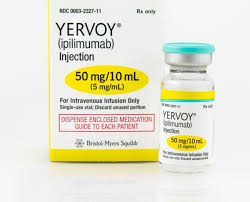
Yervoi 50mg Injection (Ipilimumab Specialized Immunotherapy Matrix)
Yervoi 50mg Injection is a high-potency pharmacological intervention containing Ipilimumab, a recombinant human monoclonal antibody specifically engineered for the clinical management of unresectable or metastatic melanoma, advanced renal cell carcinoma, and the restoration of a stabilized immunological environment. Utilizing a unique “Checkpoint-Inhibitor” delivery system, this formulation works by concurrently modulating the organized inhibition of CTLA-4 (cytotoxic T-lymphocyte-associated antigen-4) and reinforcing the structural integrity of the systemic T-cell response. By neutralizing the biochemical triggers of localized immune suppression and facilitating the organized stabilization of the anti-tumor landscape through a high-purity molecular complex, Yervoi effectively promotes a significant increase in cytotoxic T-cell activation and a reduction in systemic oncological fatigue. This targeted biochemical action provides a decisive therapeutic response by reinforcing the structural uniformity of the immune matrix while promoting a stable, physiological recovery from complex biological stressors.
Key Features
- Specifically formulated as a potent CTLA-4 blocker to target the primary pathways of T-cell regulation and tumor-induced immune evasion.
- Clinically recognized for its role in facilitating the natural optimization of the immune system’s ability to recognize and target malignant cells.
- Specifically engineered to reach therapeutic levels within the circulatory and lymphatic systems, ensuring a robust defense against localized immunosuppressive mediators.
- Acts as a functional catalyst to stabilize the internal environment and support the long-term management of advanced metastatic markers.
- Formulated as a stabilized sterile solution to address systemic exhaustion caused by chronic oncological stress factors through a high-precision delivery matrix.
Key Ingredients
- Ipilimumab (50mg/10mL)
- Tris-Hydrochloride
- Sodium Chloride
- Mannitol
- Polysorbate 80
Precautions
- This is a high-potency immunological intervention. Administration must be strictly performed by a specialist in a clinical setting following a formal oncological assessment.
- Inform your physician immediately of any changes in bowel habits, skin rashes, or endocrine symptoms, as this formulation may modulate systemic immune markers leading to immune-mediated adverse reactions.
- Regular monitoring for localized hepatic markers and liver enzyme elevations is required to maintain physiological safety during the restoration cycle.
- Strictly for intravenous infusion. Ensure the infusion rate is strictly managed to maintain the intended molecular purity and systemic tolerability.
- Discontinue use immediately and consult a specialist if you develop signs of severe colitis, pneumonitis, or acute neurological symptoms.
- Keep the vial refrigerated (2°C to 8°C). Protect from light and do not shake or freeze the vial to maintain the molecular stability of the active constituents.
How to Use
- The injection should be prepared and administered by a healthcare professional. Ensure the systemic environment is monitored for pre-infusion physiological baseline markers.
- Typically, a weight-based dose (e.g., 3 mg/kg) administered every 3 weeks constitutes the standard therapeutic dose for the restoration of physiological equilibrium.
- For optimal clinical results, administration should occur as part of a targeted cycle for the management of oncological stressors, usually spanning a 4-dose induction period.
- Administer via intravenous infusion over a period of 90 minutes to facilitate the intended molecular delivery through the systemic landscape.
- Use as a standalone restorative intervention or as part of a comprehensive combination therapy plan (e.g., with Nivolumab) as dictated by clinical requirements.
- Maintain a strict clinical schedule to document visible improvements in physiological markers and the restoration of systemic health.
Legal Disclaimer: Yervoi 50mg Injection is a specialized pharmacological product. Individual results vary based on the patient’s baseline physiological state and response. This product is for use under professional guidance only. This information is for educational purposes and is not a substitute for professional medical consultation or a formal clinical diagnosis.




Reviews
There are no reviews yet.