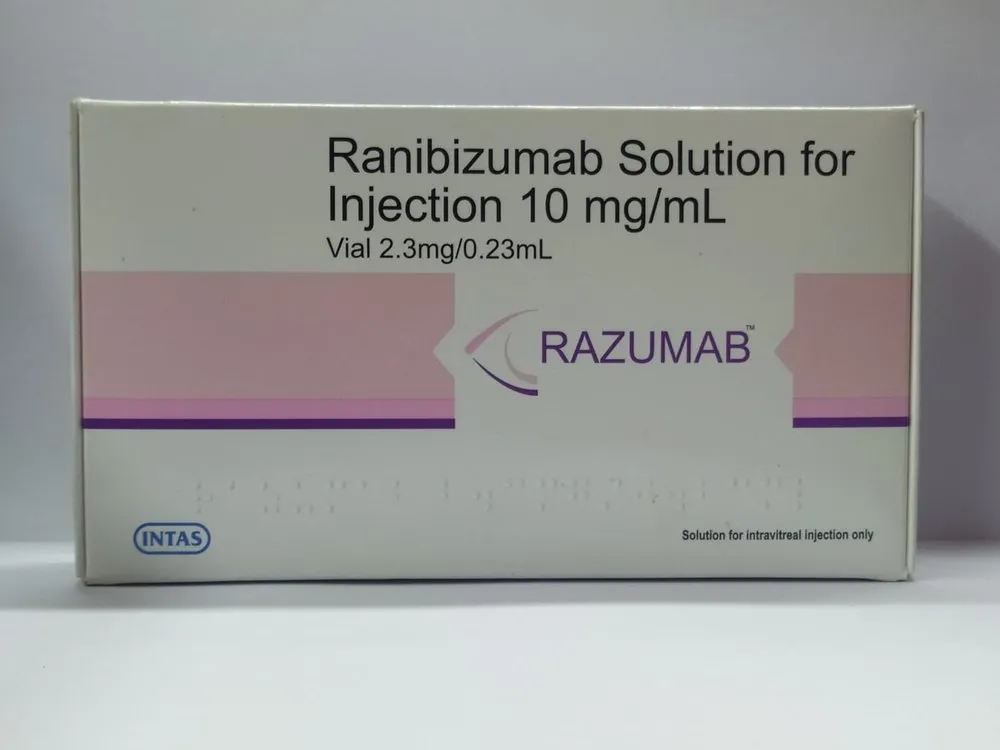
Ranibizumab 10 mg/ml (Anti-VEGF Specialized Intraocular Vascular Modulator Matrix)
Ranibizumab Solution for Injection is a high-potency pharmacological intervention containing a recombinant humanized IgG1 monoclonal antibody fragment specifically engineered for the clinical management of neovascular (wet) age-related macular degeneration (nAMD), macular edema following retinal vein occlusion (RVO), and the restoration of a stabilized intraocular environment. Utilizing a unique “Vascular-Endothelial-Growth-Factor (VEGF) Binding” delivery system, this formulation works by concurrently modulating the organized inhibition of angiogenesis and reinforcing the structural integrity of the systemic retinal response. The clinical excellence of Ranibizumab lies in its “High-Affinity-Binding” pharmacokinetic profile. Formulated for high stability and optimal receptor affinity, the active antibody fragment acts as a potent restorative agent that counteracts the physiological strain of irregular vascular proliferation and localized structural depletion of healthy retinal markers. Designed for high purity and consistent molecular delivery via a stabilized sterile solution for intravitreal injection, Ranibizumab serves as a reliable solution for achieving systemic wellness under professional ophthalmic supervision.
Key Features of Ranibizumab Solution For Injection 10 Mg/Ml
- Specifically formulated as a potent vascular modulator to target the primary pathways of intraocular neovascularization.
- Clinically recognized for its role in facilitating the natural optimization of visual acuity and the systemic management of retinal fluid accumulation.
- Specifically engineered to reach therapeutic levels within the vitreous landscape, ensuring a robust defense against localized vascular leakage.
- Acts as a functional catalyst to stabilize the internal environment and support the long-term management of stress-related retinal markers.
- Formulated as a stabilized 10 mg/ml sterile solution to address systemic exhaustion caused by chronic vascular stress factors.
Key Ingredients of Ranibizumab Solution For Injection 10 Mg/Ml
- Ranibizumab (10 mg/ml)
- α,α-Trehalose Dihydrate
- L-Histidine & Polysorbate 20
How to Use of Ranibizumab Solution For Injection 10 Mg/Ml
- Preparation: The injection should be administered as directed. Ensure the targeted ocular landscape is gently cleansed and stabilized prior to the intake of active agents.
- Dosage: Typically, application of 0.5 mg (0.05 ml) constitutes the standard therapeutic dose for the restoration of physiological equilibrium.
- Timing: For optimal clinical results, administration should occur in precise monthly or “treat-and-extend” cycles.
- Technique: Administer via intravitreal injection as per the instructional sequence provided by the ophthalmic protocol.
- Sequence: Use as the primary restorative intervention within a comprehensive retinal management plan.
- Consistency: Maintain a strict application schedule and follow the “Visual Response Tracking Protocol” to document the safe restoration of systemic health.
Precautions of Ranibizumab Solution For Injection 10 Mg/Ml
- Administration must be strictly performed by a certified ophthalmic surgeon following a formal diagnostic and retinal assessment.
- Use is strictly contraindicated in patients with active periocular or intraocular infections to maintain physiological safety.
- Strictly for intravitreal administration. Ensure the systemic environment is prepared according to the established surgical sequence.
- If both eyes require restoration, each must be treated as a separate surgical sequence to minimize the risk of cross-contamination.
- Keep vials stored in a refrigerated environment (2°C to 8°C). Protect from light; do not freeze or shake to maintain molecular stability.
Warning of Ranibizumab Solution For Injection 10 Mg/Ml
- ntravitreal injections have been associated with endophthalmitis (severe eye infection). Proper aseptic injection techniques are mandatory to maintain physiological safety.
- Acute oscillations in intraocular pressure have been documented following administration; monitoring of IOP and optic nerve perfusion is required immediately post-injection.
- There is a potential risk of arterial thromboembolic events following the systemic absorption of VEGF inhibitors; patients with a history of stroke or cardiac stress must undergo rigorous assessment.
- Patients must be monitored for localized markers of retinal tears or detachment during the restoration cycle.
- Discontinue use immediately and consult a specialist if you develop signs of acute ocular pain, severe visual oscillations, or sudden localized redness.
Legal Disclaimer: Ranibizumab Solution for Injection is a specialized pharmacological product. Individual results vary based on the patient’s baseline physiological state. This product is for use under professional guidance only. This information is for educational purposes and is not a substitute for professional medical consultation.





Reviews
There are no reviews yet.