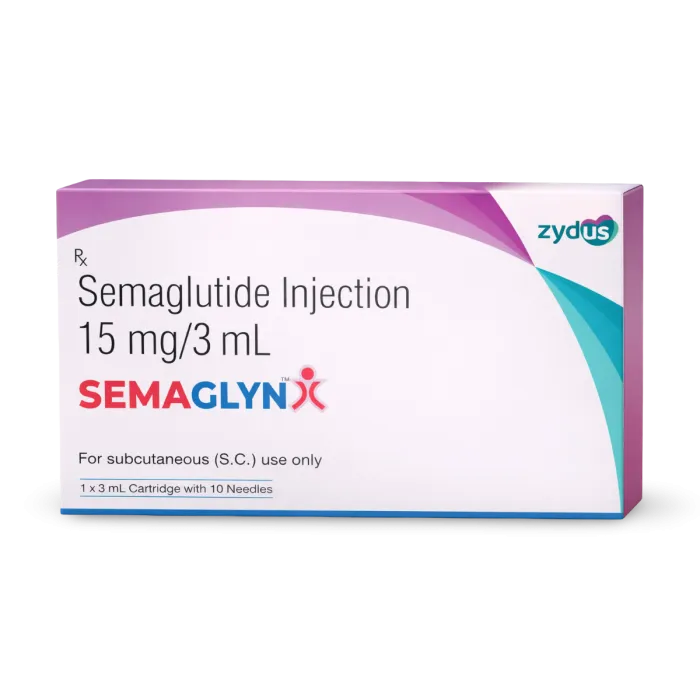
Semaglyn Injection (Semaglutide Specialized Matrix)
Semaglyn Injection is a high-potency pharmacological intervention containing Semaglutide, a long-acting glucagon-like peptide-1 (GLP-1) receptor agonist specifically engineered for the clinical management of Type 2 Diabetes Mellitus and the restoration of a stabilized metabolic environment. Utilizing a unique “Incretin-Mimetic” delivery system, this formulation works by concurrently modulating the organized secretion of insulin and reinforcing the structural integrity of the systemic glucose-regulatory axis. By neutralizing the biochemical triggers of post-prandial glycemic spikes and facilitating the organized optimization of gastric emptying and glucagon suppression through a high-purity molecular complex, Semaglyn effectively promotes a significant increase in metabolic flexibility and a reduction in systemic dullness. This targeted biochemical action provides a decisive therapeutic response by reinforcing the structural uniformity of the endocrine matrix while promoting a stable, physiological recovery from complex metabolic stressors.
Key Features
- Specifically formulated with Semaglutide to target the primary pathways of glucose-dependent insulin secretion and appetite regulation.
- Clinically recognized for its role in facilitating the natural optimization of HbA1c levels and systemic energy expenditure.
- Specifically engineered to reach therapeutic levels within the plasma, ensuring a robust defense against localized metabolic imbalances over a prolonged duration.
- Acts as a functional catalyst to stabilize the internal environment and support the intensive reduction of weight-related metabolic fatigue.
- Formulated as a stabilized solution for injection to address systemic exhaustion caused by metabolic stress factors through a high-precision delivery matrix.
Key Ingredients
- Semaglutide
- Disodium Phosphate Dihydrate
- Propylene Glycol
- Phenol
- Sterile Aqueous Vehicle
Precautions
- This is a high-potency metabolic intervention. Use must be strictly dictated by a specialist following a formal clinical diagnosis and a documented dose-escalation schedule.
- Inform your physician if you have a history of pancreatitis, as this formulation may modulate systemic markers associated with localized pancreatic inflammation.
- Regular monitoring for localized metabolic dullness, nausea, or altered gastric emptying is required to manage secondary responses during the maintenance phase.
- Strictly for subcutaneous injection in the abdomen, thigh, or upper arm. Do not administer intravenously or intramuscularly to ensure the intended physiological response.
- Discontinue use immediately if persistent localized cutaneous eruptions, severe abdominal discomfort, or signs of an acute hypersensitivity reaction occur.
- Store in a refrigerated environment (2°C to 8°C). Protect from direct light and do not freeze to maintain the molecular stability of the active constituents.
How to Use
- The solution should be clear and colorless. Ensure the delivery equipment is ready and the epidermal surface at the injection site is cleansed for the intake of active agents.
- Typically, one weekly injection constitutes the standard therapeutic dose for the restoration of physiological equilibrium, as dictated by clinical requirements.
- For optimal clinical results, administration should occur on the same day each week, at any time of day, as part of your primary wellness regimen.
- Administered via subcutaneous injection. Rotate the injection site weekly to maintain the structural integrity of the subcutaneous tissue and support absorption.
- Use as a standalone restorative intervention or as part of a comprehensive metabolic plan including nutritional modifications as dictated by clinical requirements.
- Maintain a regular application schedule to document visible improvements in physiological markers and the restoration of systemic health.
Legal Disclaimer: Semaglyn Injection is a specialized pharmacological product. Individual results vary based on the patient’s baseline metabolic state and response. This product is for use under professional guidance only. This information is for educational purposes and is not a substitute for professional medical consultation or a formal clinical diagnosis.






Reviews
There are no reviews yet.